Are you searching for the Estimation of protein by the Bradford method protocol? The protocol is given below. The Bradford assay is very fast and uses about the same amount of protein as the Lowry assay. It is fairly accurate and samples that are out of range can be retested within minutes. The Bradford is recommended for general use, especially for determining the protein content of cell fractions and assessing protein concentrations for gel electrophoresis.

Assay materials including color reagent, protein standard, and instruction booklet are available from Bio-Rad Corporation.
The method described below is for a 100 µl sample volume using a 5 ml color reagent. It is sensitive to about 5 to 200 micrograms protein, depending on the dye quality. In assays using 5 ml color reagent prepared in the lab, the sensitive range is closer to 5 to 100 µg protein.
The Bradford protein assay was developed by Marion M. Bradford in 1976. It is a quick and accurate spectroscopic analytical procedure used to measure the concentration of protein in a solution. The reaction is dependent on the amino acid composition of the measured proteins.
Scale down the volume for the “microassay procedure,” which uses 1 ml cuvettes. Protocols, including the use of microtiter plates, are described in the flyer that comes with the Bio-Rad kit.
Estimation of Protein by Bradford method
Materials
- Lyophilized bovine plasma gamma globulin or Bovine Serum Albumin (BSA)
- Coomasie Brilliant Blue 1
- 0.15 M NaCl
- Spectrophotometer and tubes
- Micropipettes
PROCEDURE (STANDARD ASSAY, 20-150 µg protein; 200-1500 µ g/ml)
- Prepare a series of protein standards using BSA diluted with 0.15 M NaCl to final concentrations of 0 (blank = NaCl only), 250, 500, 750 and 1500 µ g BSA/ml. Also, prepare serial dilutions of the unknown sample to be measured.
- Add 100 µ l of each of the above to a separate test tube (or spectrophotometer tube if using a Spec 20).
- Add 5.0 ml of Coomasie Blue to each tube and mix by vortex, or inversion.
- Adjust the spectrophotometer to a wavelength of 595 nm, and blank using the tube from step 3 which contains 0 BSA.
- Wait 5 minutes and read each of the standards and each of the samples at 595 nm wavelength.
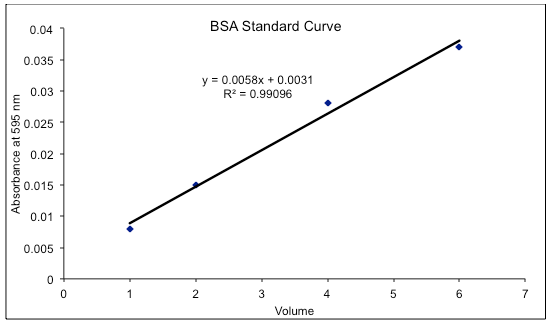
- Plot the absorbance of the standards vs their concentration. Compute the extinction coefficient and calculate the concentrations of the unknown samples.
PROCEDURE (MICRO ASSAY, 1-10 µ g protein)
- Prepare standard concentrations of BSA of 1, 5, 7.5 and 10 µ g/ml. Prepare a blank of NaCl only. Prepare a series of sample dilutions.
- Add 100 µ l of each of the above to separate tubes (use micro-centrifuge tubes) and add 1.0 ml of Coomasie Blue to each tube.
- Turn on and adjust a spectrophotometer to a wavelength of 595 nm, and blank the spectrophotometer using 1.5 ml cuvettes.
- Wait 2 minutes and read the absorbance of each standard and sample at 595 nm.
- Plot the absorbance of the standards vs their concentration. Compute the extinction coefficient and calculate the concentrations of the unknown samples.
Analysis
Prepare a standard curve of absorbance versus micrograms protein and determine amounts from the curve. Determine concentrations of original samples from the amount of protein, volume/sample, and dilution factor, if any.
Comments
The dye reagent reacts primarily with arginine residues and less so with histidine, lysine, tyrosine, tryptophan, and phenylalanine residues. Obviously, the assay is less accurate for basic or acidic proteins. The Bradford assay is rather sensitive to bovine serum albumin, more so than “average” proteins, by about a factor of two. Immunoglobulin.G (IgG – gamma globulin) is the preferred protein standard. The addition of 1 M NaOH was suggested by Stoscheck (1990) to allow the solubilization of membrane proteins and reduce the protein-to-protein variation in color yield.
References
- Bradford, MM. A rapid and sensitive for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72: 248-254. 1976.
- Stoscheck, CM. Quantitation of Protein. Methods in Enzymology 182: 50-69 (1990).
Few other Protocols
- Effect of Temperature on Amylase activity
- Isolation of Glycogen from Liver
- Estimation of Pyruvate by using 2,4 DNP
- Effect of Enzyme Concentration on Alpha-Amylase Activity
- Isolation of Starch from Potatoes
- Estimation of Saponification Value of Fats/Oils
- Time Course of Enzyme Activity (Alpha-Amylase)
- Effect of Substrate Concentration on Alpha-Amylase Activity
- Effect of pH on enzyme activity and determination of optimum pH
- Estimation of Tyrosine by the “FC Method”
- Estimation of Proteins by “Lowry method”
- Estimation of Protein by the Bradford Method
