The endocrine system comprises glands that produce hormones and chemical messengers coordinating bodily functions. Hormones regulate growth, metabolism, reproduction, mood, and tissue function. This article will provide an in-depth overview of the Mechanisms of Hormone Action and the endocrine system.
What Are Hormones and How Do They Work?
Hormones are substances secreted by the endocrine glands that travel through the bloodstream to target distant organs and tissues. Once hormones reach their target cells, they bind to specific receptors on or inside cells to trigger responses and regulate growth, development, metabolism, reproduction, mood, etc.
Some key points about hormones:
- Hormones act slowly and cause prolonged responses.
- Very small amounts of hormones can have big effects.
- Hormones work through feedback mechanisms that regulate their release.
- Imbalances in hormones can cause various disorders.
There are three main classifications of hormones:
- Peptide/protein hormones like insulin, growth hormone, and oxytocin.
- Steroid hormones like testosterone, progesterone, and estrogen.
- Amine hormones like epinephrine, norepinephrine, and thyroxine.
Mechanisms of Hormone Action: How Hormones are working
Mechanisms of Hormone Action at the cellular level begins with the association of the Hormone and its specific receptor. Hormones can be classified by the nature of the signal (or) second messenger used to mediate hormonal action within the cell. Several of these second messengers have been defined, but the intracellular signal has been discovered for several hormones.
Major hormones of the human endocrine system like insulin are water soluble so they bind cell membrane hormone receptors. Steroid hormones are derived from cholesterol while peptide hormones are derived from amino acids.
When the hormone binds to its receptor it induces a conformational change that activates the receptor to trigger a cellular response. Examples of hormones that use the second messenger system include epinephrine and glucagon.
The receptor becomes activated when the hormone binds, triggering second messenger pathways specific to that hormone’s receptor. There are many different hormones like insulin, growth hormone and oxytocin that exert different effects by binding receptors.
Based on the mechanism of Hormonal action, the hormones may be classified into two types:
A. Peptide Hormonal Action and its mechanism:
There are two main types of hormone receptors: intracellular hormone receptors found inside the cell and cell membrane hormone receptors that bind hormones at the cell surface. Lipid soluble hormones like steroid hormones are able to diffuse across the membrane to reach intracellular receptors.
1. Cyclic AMP:
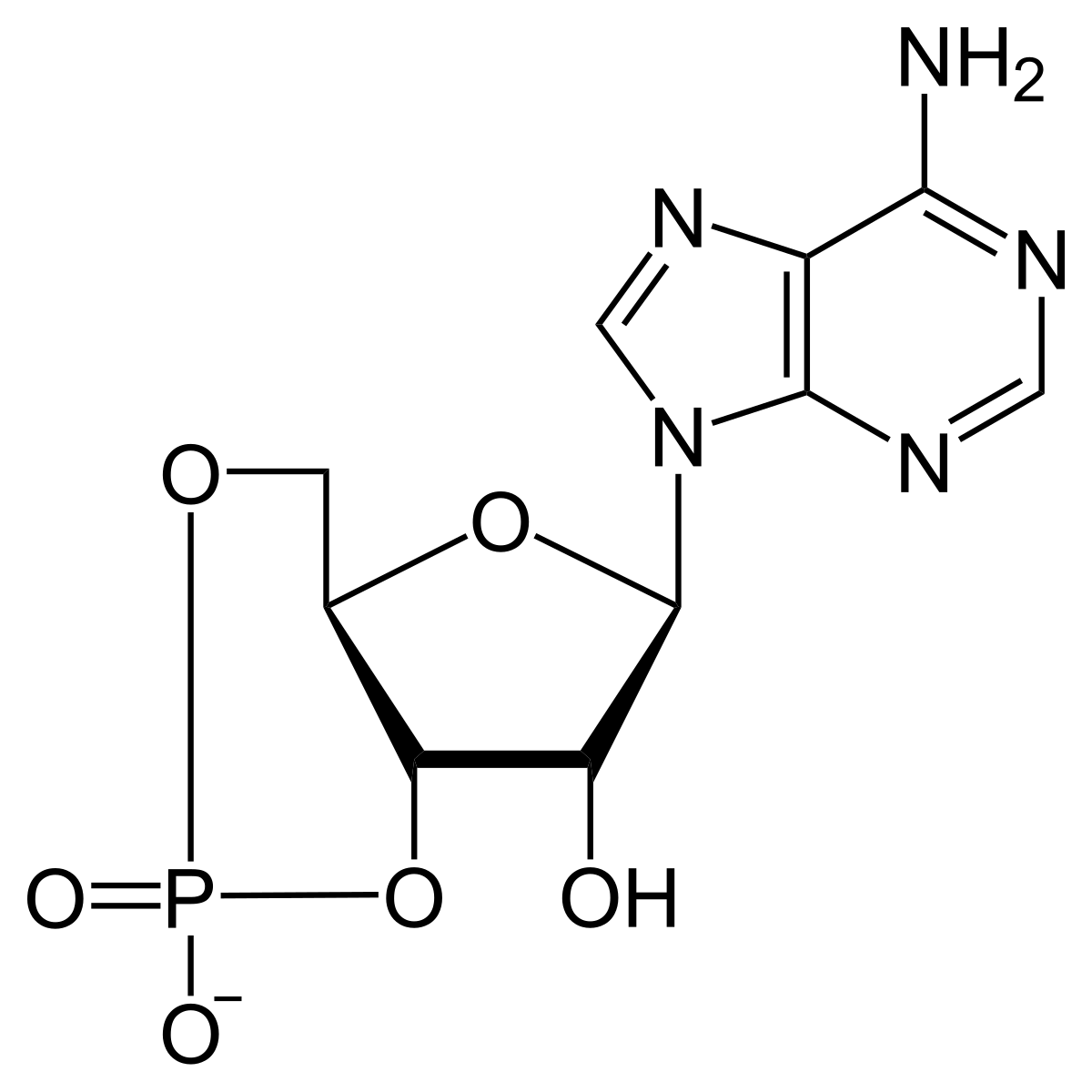
- Cyclic AMP was first discovered by “Earl.W.Sutherland” & “T.W.Rall” in 1960, who was awarded the Nobel Prize in 1971.
- “Adenyl cyclase(AC)” converts ATP to cAMP (3′,5’-Cyclic AMP), and “Phosphodiesterase” hydrolyzes cAMP to 5’AMP.
- Cyclic AMP acts as an intracellular hormonal mediator.
- The cAMP is also frequently called the Second messenger for hormone mediation, the first messenger being the original Hormone.
2. G-Protein (or) Nucleotide Regulatory Protein:
- “Alfred G. Gilman” and “Martin Rodbell” were awarded the Nobel Prize in 1994 for their work on G-protein.
- The G-protein is a peripheral membrane protein consisting of alpha, beta, and gamma subunits.
- When GTP is attached, it becomes active. Then, it can activate the “Adenylate cyclase”.
3. G-Protein activates Adenylate Cyclase:
When the Hormone–receptor complex is formed, the activated receptor stimulates the G-protein, which carries the excitation signal to adenylate cyclase. This signal transduction takes place in the plasma membrane. The adenylate cyclase is embedded in the plasma membrane.
Mechanisms of Hormone Action:
The extracellular messenger, the Hormone (H), combines with the specific receptor (R) on the plasma membrane. The H-R complex activates the regulatory component of the protein designated as G-protein (or) nucleotide regulatory protein.
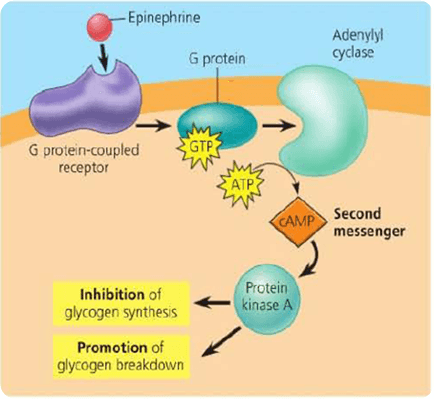
G-proteins are so named because they are bound to GTP. The binding of hormones to the receptor triggers the exchange of GTP for bound GDP. The H-R binds to the G-protein, induces the release of bound GDP & allows GTP to bind. The alpha subunit binds the GTP and dissociates from the beta-gamma subunits.
The G-alpha-GTP activates the adenylate cyclase (AC) exposed to the cytoplasm, causing immediate conversion of cytoplasmic ATP into cAMP. The cAMP then acts inside the cell to initiate several cellular functions before it is destroyed.
The cAMP elicits many of its effects by activating protein kinases. These kinases are tetrameric molecules with two regulatory (R) and two catalytic subunits (R2C2). This complex has no activity. But cAMP binds to the regulatory and catalytic subunit and catalytic subunits.
The catalytic subunit transfers a phosphate group from ATP to different enzyme proteins. Phosphorylation usually occurs on the –OH group Serine, Threonine (or) Tyrosine residues of the substrates. The enzymes may be activated (or) inactivated by this phosphorylation.
The second messenger system relies on cyclic AMP to relay the hormone response after the protein hormone binds its membrane receptor. Nuclear hormone receptors like those for steroid and thyroid hormones directly trigger gene transcription once the hormone binds.
The follicle-stimulating hormone binds to receptors on ovarian follicles to stimulate maturation of ova and secretion of estrogen. The number of hormone receptors present affects the cell’s sensitivity and response to a given hormone.
Some important hormone-responsive protein kinases:
- Calmodulin-dependent kinase
- cAMP-dependent kinases-I & II
- cAMP-dependent kinase
- Epidermal growth factor-dependent tyrosine kinase
- Insulin-dependent tyrosine kinase
- Pyruvate dehydrogenase kinase
- Phosphorylase kinase
Other actions of cAMP are summarized below:
- cAMP helps in the conversion of cholesterol in pregnenolone.
- It stimulates 11-beta hydroxylase for catecholamine synthesis.
- It stimulates tyrosine hydroxylase for catecholamine synthesis.
- It stimulates melatonin synthesis by increasing the N-acetyltransferase of the pineal gland.
- It causes the release of insulin from pancreatic islet cells.
- Histamine stimulates the production of gastric HCl through cAMP.
- It induces cells to differentiate.
Mechanism of action of certain toxins:
a) Cholera:
- The toxin binds to a ganglioside on the intestinal mucosal cell, which leads to ribosylation of the alpha subunit of Gs-Protein.
- This results in inhibiting the inherent GTPase activity and irreversible activation of G-Protein.
- Therefore, Adenylate cyclase remains active and keeps the cAMP level high.
- It prevents absorption of salts from the intestine, leading to watery diarrhea and loss of water from the body.
b) Pertussis:
- The toxin irreversibly activates adenylate cyclase by promoting the ADP ribosylation of G-ai, preventing the G-aI subunit from activating.
c) NaF:
- It is another irreversible activator of cyclase.
b. Steroid Hormone Action and its Mechanism
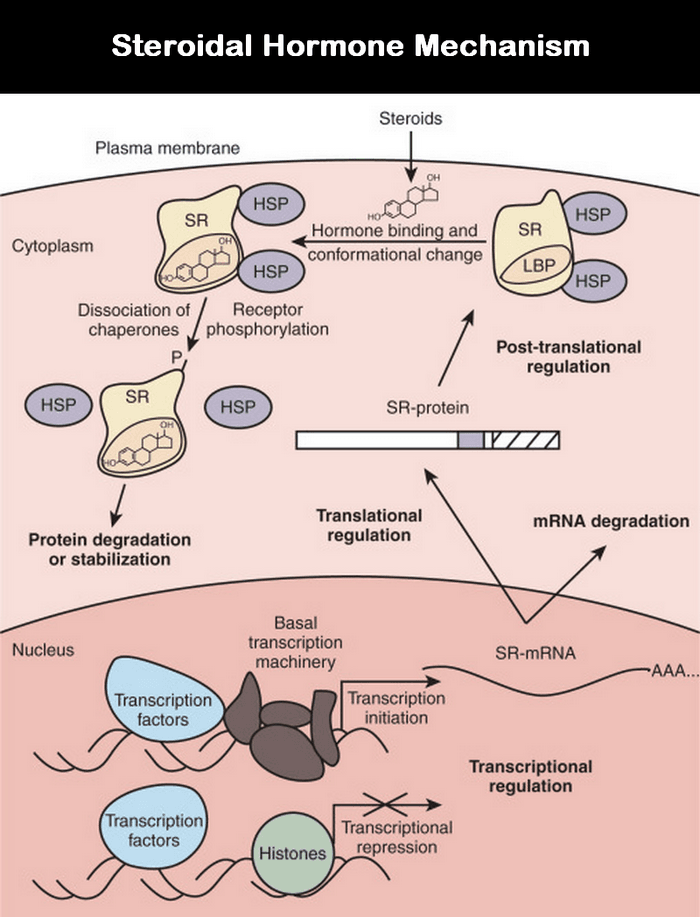
Hormones can’t pass through the plasma membrane, so binding of the hormone to its receptor triggers the cell’s response. The hormone that binds and activates the receptor is specific to a given cell’s receptor for that particular hormone.
The hormones in this group include steroidal hormones and thyroid hormones. They diffuse through the plasma membrane and bind to the receptors in the cytoplasm. The Hormone – Receptor (HR) complex is formed.
The complex is then translocated to the nucleolus. The HR binds to the DNA’s promoter (or) regulatory elements and activates (or) inactivates genes.
The Hormone binds to the specific area of the genes, referred to as the “Hormone Responsive Element (HRE).” Steroid hormone receptor proteins have a molecular weight of about 80 to 100 KD. Each monomer binds to a single steroid molecule at a hydrophobic site, but on binding to genes, they dimerize.
The receptors bind by their DNA binding capacity. The steroid-regulated genes have “Steroid receptor binding sites” (or) “Steroid response elements (SREs).”
The SRE acts as an enhancer element and, when stimulated by hormones, would increase transcriptional activity. Binding to the SRE sequence leads to dimerization of the receptor.
The sequence of events is as follows:
- The steroidal Hormone enters the cytoplasm of the target cell, where it binds with a specific high-affinity receptor protein.
- The receptor protein-hormone complex, so formed, diffuses into (or is transported into) the nucleolus, which reacts with the nuclear chromatin.
- Some along this route, the receptor protein is structurally altered to form a smaller protein with low molecular weight (or) else the steroid hormone is transferred to a second smaller protein.
- The combination of the small protein and Hormone is now the active factor that stimulates the specific genes to form mRNA in the nucleolus.
- The mRNA diffuses into the cytoplasm, accelerating the translocation process at the ribosome to synthesize new proteins.
Points to remember:
- Hormones of the human body regulate various physiological processes.
- A hormone response element is a crucial part of gene regulation.
- Two hormones work together to maintain homeostasis.
- Cellular changes occur upon hormone binding to their receptors.
- Each specific hormone has a unique function in the body.
- The response of a hormone depends on its concentration.
- Binding of the hormone activates specific signaling pathways.
- Receptors are specific to a given hormone type.
- Water-soluble hormones easily dissolve in bodily fluids.
- Hormones derived from amino acids include epinephrine and thyroxine.
- The effects of hormones can be widespread and diverse.
- Signaling pathways depend on the type of mechanism involved.
- The hormone is called by its specific chemical name.
- The cAMP second messenger system is essential for many hormones.
- Inhibition of a hormone’s action can lead to health problems.
- Hormones are secreted by various glands in the body.
- Chemical messengers, hormones transmit signals between cells.
- Water-soluble hormone binds to cell membrane receptors.
- To exert their effects, hormones must bind to target cells.
- Excessive hormone levels can disrupt normal bodily functions.
- Hydrophobic hormones diffuse through cell membranes to reach their targets.
The Endocrine System and Its Hormone-Producing Glands
The endocrine system refers to the glands of the body that secrete hormones directly into the bloodstream to be transported to distant target organs. The major glands of the endocrine system include:
- Hypothalamus: Located in the brain, this gland secretes hormones that control other endocrine glands. Key hormones: Releasing & inhibiting hormones.
- Pituitary: The “master gland” that secretes hormones controlling growth, metabolism, and reproduction. Key hormones: Growth hormone, TSH, ACTH, FSH, LH.
- Thyroid: Secretes hormones that control metabolism and growth. Key Hormone: Thyroxine (T4).
- Parathyroids: Secrete parathyroid Hormone that controls blood calcium levels.
- Adrenal: Secrete hormones like cortisol that control the stress response.
- Pancreas: Secretes insulin and glucagon that regulate blood glucose levels.
- Ovaries & Testes: Secrete sex hormones like estrogen, progesterone, and testosterone.
Hormone Regulation Through Feedback Mechanisms
To maintain homeostasis, the endocrine system uses feedback mechanisms that control hormone levels through negative and positive feedback:
- Negative feedback decreases hormone production when levels get too high.
- Positive feedback increases hormone production when levels are too low.
For example, the thyroid gland secretes thyroid hormones (T3 & T4) that increase metabolism. If T3/T4 levels get too high, this triggers negative feedback to the pituitary and hypothalamus, decreasing TSH release and slowing thyroid hormone production.
How Do Hormones Interact With Receptors to Trigger Responses?
Hormone receptors are proteins located inside or on the surface of cells that specifically bind to hormones to trigger cellular responses. The key steps in hormone-receptor interactions are:
- The Hormone is released into the bloodstream by an endocrine gland.
- Hormone travels through circulation and reaches the target tissue/organ.
- Hormone binds to matching receptors on cells.
- Receptor binding activates intracellular enzymes and pathways.
- The cell responds by changing enzyme activity, ion concentrations, protein synthesis, etc.
- Physiological responses like growth, metabolism, mood, and reproduction occur in target tissues.
Common Endocrine System Disorders
Imbalances in hormone levels can occur from gland disorders, nodules, autoimmunity, etc. Common endocrine disorders include:
- Diabetes mellitus: Insufficient insulin leads to high blood glucose.
- Thyroid disorders: Hyper/hypothyroidism from too much/little thyroid hormone.
- Addison’s disease: Adrenal insufficiency with low cortisol levels.
- Cushing’s syndrome: Excess cortisol production by adrenal glands.
- Hyper/hypopituitarism: Over/underactive pituitary gland.
- Polycystic ovarian syndrome (PCOS): Excess androgen and irregular ovulation.
Endocrine disorders have varied symptoms but may include fatigue, weight changes, irritability, reproductive issues, slow growth, and bone problems. Treatment depends on the specific condition but may involve hormone replacement, medications, surgery, and lifestyle changes.
The Hypothalamic-Pituitary Axis Controls Endocrine Glands
The hypothalamus and pituitary gland comprise the hypothalamic-pituitary axis that links the nervous system to the endocrine system. The hypothalamus produces releasing and inhibiting hormones that control pituitary hormone secretion. The pituitary, in turn, regulates downstream endocrine glands.
Key hypothalamic hormones:
- GnRH – Stimulates FSH/LH release
- TRH – Stimulates TSH release
- CRH – Stimulates ACTH release
- Dopamine – Inhibits prolactin release
Key pituitary hormones:
- Growth hormone (GH)
- Thyroid-stimulating Hormone (TSH)
- Adrenocorticotropic hormone (ACTH)
- Follicle stimulating Hormone (FSH)
- Luteinizing Hormone (LH)
- Prolactin (PRL)
This axis allows the brain to control growth, metabolism, stress, reproduction, lactation, and other processes through the endocrine system.
The Role of Hormones in Growth and Development
Hormones drive growth and development at different life stages:
- Childhood growth: Growth hormone stimulates the growth of bones and tissues. Thyroid hormones also impact growth rate.
- Puberty: The hypothalamus activates the pituitary to secrete FSH/LH, which stimulates sex hormone secretion and leads to secondary sexual characteristics.
- Menstrual cycle: Estrogen and progesterone levels fluctuate to control the menstrual cycle, preparing for possible pregnancy each month.
- Pregnancy/lactation: HCG, estrogen, and progesterone support pregnancy. Prolactin stimulates milk production for breastfeeding.
- Aging: Declining growth and sex hormone levels with age lead to changes like slowed metabolism and bone loss.
How Hormones Respond to Stress
The hypothalamic-pituitary-adrenal (HPA) axis controls the stress response:
- Stress exposure (injury, threat) activates the hypothalamus.
- The hypothalamus secretes the CRH hormone, which triggers the pituitary.
- The pituitary releases ACTH, which stimulates the adrenal cortex.
- The adrenal cortex secretes cortisol, the main human stress hormone.
- Cortisol then increases blood sugar, suppresses inflammation, and counter stress.
However, chronic stress can dysregulate the HPA axis, causing excessive cortisol and symptoms like fatigue, anxiety, high blood pressure, and weight gain.
The Impact of Hormones on Behavior and Mental Health
Hormones like cortisol, estrogen, testosterone, oxytocin, vasopressin, and dopamine can influence mood, cognition, motivation, attention, aggression, bonding, and other behaviors.
- Estrogen and serotonin influence mood and depression risk in women.
- Testosterone impacts dominance, aggression, and risk-taking.
- Cortisol levels affect the stress response and anxiety.
- Oxytocin promotes social bonding.
- Dopamine regulates motivation, attention, and addiction.
Hormonal imbalances may contribute to conditions like depression, anxiety, schizophrenia, ADHD, autism, and aggressive or addictive behaviors. More research is needed into the hormone-behavior links.
Ongoing Research and Future Directions in Endocrinology
Active areas of research in endocrinology include:
- Developing new hormone-based therapies for diabetes, infertility, hormone-responsive cancers, osteoporosis, and obesity.
- Understanding hormone-behavior connections and using hormone treatments for psychiatric and neurological conditions.
- Investigating links between endocrine disruptors like BPA and health problems.
- Studying hormone impacts on aging and age-related diseases.
- Using stem cells to generate hormone-producing cells for replacement therapy.
- Creating artificial smart insulin delivery systems.
- Interactions between the endocrine system, nervous system, and immune system.
- Precision medicine approaches are based on individuals’ genetic predispositions.
Frequently Asked Questions
What are the main functions of hormones in the human body?
Hormones have many important functions, including regulating growth and development, metabolism, reproduction, mood, tissue function, stress response, sexual function, and other essential physiological processes. The endocrine system uses complex hormonal communications to coordinate and control bodily homeostasis.
How quickly do hormones take effect in the body?
Unlike neurotransmitters used for quick nerve impulses, hormones circulate through the bloodstream to elicit slower, prolonged, cascading responses in target tissues that can take hours, days, or weeks to exert their full effect. However, hormones bind to receptors rapidly, within seconds to minutes.
Do hormones impact behavior and personality?
Growing research shows that hormones like oxytocin, estrogen, cortisol, testosterone, dopamine, and others can influence human behavior, mood, cognition, motivation, attention, social bonding, aggressiveness, and mental health conditions. More studies are needed to understand these effects fully.
What are the most common endocrine disorders?
Some of the most prevalent endocrine system disorders include diabetes (pancreas), thyroid disorders like hyper/hypothyroidism, adrenal insufficiency (Addison’s disease), Cushing’s syndrome, pituitary tumors or hyper/hypopituitarism, and polycystic ovarian syndrome (PCOS) in women.
How are hormones monitored and tested?
Doctors can check hormone levels through simple blood tests, urine tests, or other procedures like glucose tolerance tests or thyroid-stimulating hormone assessments. Results are compared to normal reference ranges to check for deficiencies or excess. Testing helps diagnose endocrine problems.
Conclusion: Mechanism of Action and types of hormones
In summary, hormones are crucial chemical messengers regulating nearly all major bodily processes. The endocrine system carefully controls complex hormonal release and interactions to maintain homeostasis.
A hormone is a chemical messenger released by endocrine glands that travels through the blood to act on distant target cells. Hormones such as steroid hormones, peptide hormones and amine hormones play important regulatory roles.
Hormonal imbalances can occur for various reasons, leading to endocrine disorders with diverse symptoms. Research continues to uncover new insights into the Mechanisms of Hormone Action that may lead to improved diagnostic tests, treatments, and synthetic hormones. Awareness of your body’s hormonal state and discussing imbalances with your doctor are key to optimizing health.
Additional Points:
- The main types of hormone mechanism include the second messenger system, hormone binding nuclear receptors, or direct activation of enzyme activity. A given hormone can bind and activate only receptors specific to that hormone.
- Cyclic AMP second messenger system amplifies the response to hormone binding by activating protein kinases that phosphorylate enzymes. Some receptors induce release of ions as a second messenger when activated by a hormone.
- Inhibition of a hormone’s effects can occur by preventing its synthesis or release, blocking its binding to receptors, or interfering with post-receptor signaling. Hormones are the chemical messengers that mediate inter-cellular communication by binding receptors.
- It’s important to understand the mechanisms of hormone action through receptor binding and signal transduction when learning about the hormone receptor complex. Lipid soluble hormones like steroids can directly pass through the membrane to bind intracellular receptors.
- Once the hormone binds to the target receptor, it induces a conformational change leading to activation of effector proteins and second messengers that release various hormones. The amount of hormone secreted and circulating determines the overall effect on the target cell.
- Reproductive hormones testosterone and estrogen, the lipid-derived hormone cortisol, and water-soluble hormone insulin work via different mechanisms. Peptide hormones must first bind cell membrane receptors while steroid hormones can enter the cell.
- Growth hormone–inhibiting hormone somatostatin and growth hormone–releasing hormone control human growth hormone release. Maintaining proper hormone levels is crucial, as excessive amounts can disrupt homeostasis.
- Feedback loops ensure original hormone release is eventually inhibited to prevent over-stimulation. Amine hormones originate from the amino acid tyrosine and contain nitrogen. The hydrophobic steroid hormone diffuses directly into cells.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.