Nucleic acids are organic polymers, composed of monomer units known as nucleotides. Nucleotides are energy-rich compounds that drive metabolic processes in all cells. They also serve as chemical signals, key links in cellular systems that respond to hormones and other extracellular stimuli, and are structural components of a number of enzyme cofactors and metabolic intermediates.

There are very few different types of nucleotides. The main functions of nucleotides are information storage (DNA), protein synthesis (RNA), and energy transfers (ATP and NAD).
- What is RNase? What is the importance of living things?
- Why Peptide bonds are Backbone of the Proteins
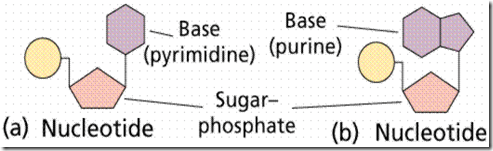
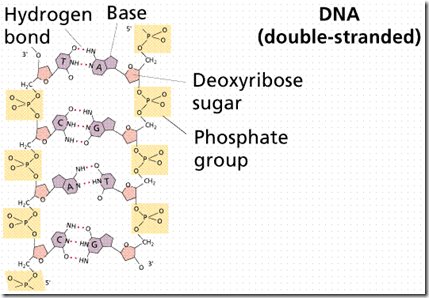
Nucleotides (Backbone unit of Nucleic Acids), shown in Figure 1, consist of a sugar, a nitrogenous base, and a phosphate. The sugars are either ribose or deoxyribose. They differ by the lack of one oxygen in deoxyribose. Both are pentoses usually in a ring form.
The back structure of Nucleic Acids
The nucleic acids (DNA and RNA) are the molecular repositories for genetic information and are jointly referred to as the ‘molecules of hereditary’. The structure of every protein, and ultimately of every cell constituent, is a product of information programmed into the nucleotide sequence of a cell’s nucleic acids.
There are five nitrogenous bases. Purines (Adenine and Guanine) are double-ring structures, while pyrimidines (Cytosine, Thymine, and Uracil) are single-ringed.
Deoxyribonucleic acid (better known as DNA) is the physical carrier of inheritance for 99% of living organisms.
DNA functions in information storage. The English alphabet has 26 letters that can be variously combined to form over 50,000 words. DNA has four letters (C, G, A, and T, the nitrogenous bases) that code for twenty words (the twenty amino acids found in all living things) that can make an infinite variety of sentences (polypeptides). Changes in the sequences of these bases information can alter the meaning of a sentence.
For example take the sentence: I saw Elvis. This implies certain knowledge (that I’ve been out in the sun too long without a hat, etc.).
If we alter the sentence by inverting the middle word, we get: I was Elvis (thank you, thank you very much). Now we have greatly altered the information.
A third alteration will change the meaning: I was Levis. Clearly the original sentence’s meaning is now greatly changed.
Changes in DNA information will be translated into changes in the primary structure of a polypeptide, and from there to the secondary and tertiary structures.
What Are Mutations?
A mutation is any change in the DNA base sequence. Most mutations are harmful, few are neutral, and a very few are beneficial and contribute to the organism’s reproductive success. Mutations are the wellspring of variation, variation is central to Darwin and Wallace’s theory of evolution by natural selection. Check the details on Mutations and its types
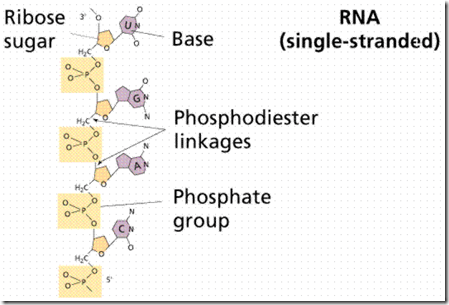
Ribonucleic acid (RNA)
RNA was discovered after DNA. DNA, with exceptions in chloroplasts and mitochondria, is restricted to the nucleus (in eukaryotes, the nucleoid region in prokaryotes). RNA occurs in the nucleus as well as in the cytoplasm (also remember that it occurs as part of the ribosomes that line the rough endoplasmic reticulum). There are three types of RNA:
- Messenger RNA (mRNA) is the blueprint for the construction of a protein.
- Ribosomal RNA (rRNA) is the construction site where the protein is made.
- Transfer RNA (tRNA) is the truck delivering the proper amino acid to the site at the right time.
Adenosine triphosphate, better known as ATP (Figure 25), the energy currency or coin of the cell, transfers energy from chemical bonds to endergonic (energy absorbing) reactions within the cell. Structurally, ATP consists of the adenine nucleotide (ribose sugar, adenine base, and phosphate group, PO4-2) plus two other phosphate groups.
- Chemiosmotic Theory by ATP Synthase Complex
- Electron Transport Chain Mechanism in Mitochondria
- Oxidative Phosphorylation: Fate of Electrons in Mitochondria
- Complete Guide on Intermediary Metabolism and its types
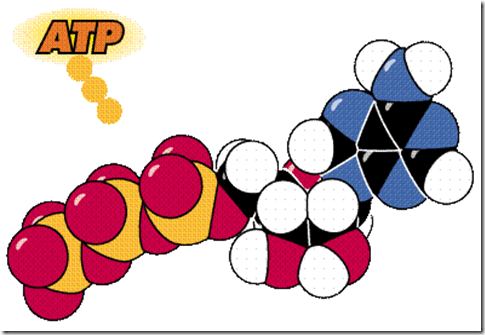
Energy is stored in the covalent bonds between phosphates, with the greatest amount of energy (approximately 7 kcal/mole) in the bond between the second and third phosphate groups. This covalent bond is known as a pyrophosphate bond.
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.