Pyrimidine synthesis is similar to that of purines (purine synthesis). In the de-novo synthesis of pyrimidines, the ring is synthesized first, and then the pyrimidine ring is tacked onto the heterocyclic ring. Thus, the same amido-alkylation reaction between the appropriately substituted ketone and the nitrogen of ammonium chloride is also used to synthesize the pyrimidine.

The pyrimidine biosynthesis (denovo pyrimidine synthesis pathway) was first observed in mutants of the bread mole Neurospora crassa, which cannot synthesize pyrimidine and they require cytosine and cytosine Uracil in their growth medium.
Source of Atoms in Pyrimidine Ring
Pyrimidines include cytosine, uracil, and thymine, which are all part of the chemical bases of DNA and RNA. Pyrimidines are usually found in nucleotides, nucleic acids, cytidine triphosphates, ribonucleosides, and ribonucleotides. The pyrimidine ring is formed using an amine and nitrogen with a three-atom ring.
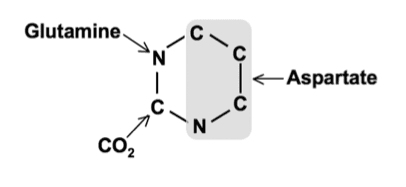
- N1, C4, C5, and C6 → Comes from Aspartate
- N3 → Comes from Glutamine
- C2 → Comes from HCO3– (Carbonic acid)
Steps of Pyrimidine Synthesis
The pyrimidine synthesis pathway is responsible for synthesizing uracil nucleotides from cytosine and thymine nucleotide precursors. This process occurs in prokaryotes, eukaryotes, and archaea. It also occurs during DNA replication and the repair of damaged or incomplete replicated strands of DNA.
It is one of the nucleic acid synthesis pathways. In the Pyrimidines, there are three Nucleotide molecules: UTP, CTP, and TTP. The following steps can explain the Denovo pyrimidine synthesis pathway.
- Synthesis of Carbamoyl Phosphate
- Synthesis of Carbamoyl Aspartate
- Ring Closure to form dihydroorotate
- Oxidation of Dihydro Orotate
- Addition of ribose Phosphate moiety
- Decarboxylation to form UMP
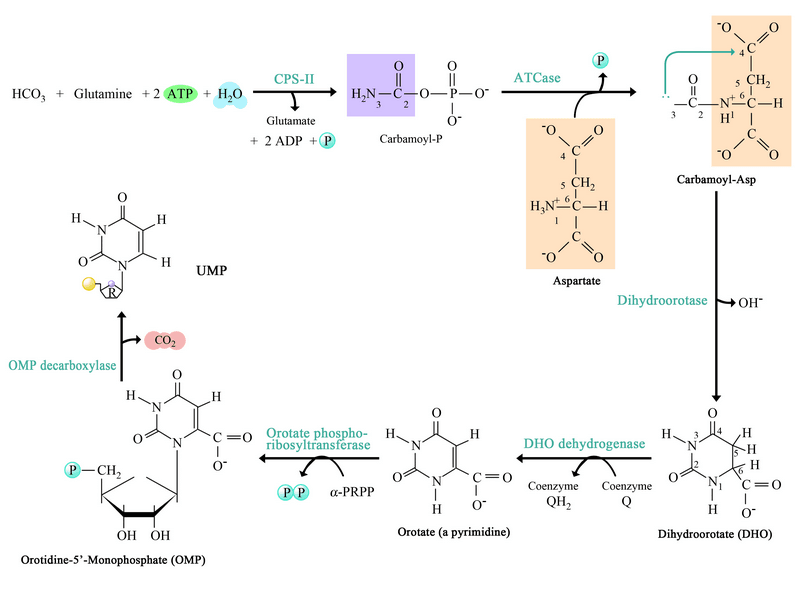
Step 1: Synthesis of Carbamoyl Phosphate
- The first reaction of pyrimidine synthesis is the synthesis of carbamoyl phosphate by utilizing the amide form of glutamine (glutamate) and HCO3-(carbonic acid).
- This reaction is catalyzed by carbamoyl phosphate synthetase-II. The enzyme is a cytosolic enzyme.
- In this reaction, two ATP molecules are consumed.
HCO3– (Carbonic acid) + Glutamine (Gln) → Carbamoyl Phosphate + 2 AMP
Step 2: Synthesis Carbamoyl Aspartate
- Carbamoyl phosphate is condensing with Aspartic acid it forms carbamoyl aspartate is catalyzed by Aspartate Transcarbamoylase (ATCase).
Carbamoyl Phosphate + Aspartate → Carbamoyl Aspartate + H3PO4
Step 3: Ring Closure to form dihydroorotate:
- Carbamoyl Aspartate is converted into Dihydro Orotate by ring closure mechanism.
- This reaction is catalyzed by Dihydro Orotase.
Carbamoyl Aspartate → Dihydro Orotate + H20
Step 4: Oxidation of dihydroorotate
- The hydro orotate is irreversibly oxidized to orotate by the enzyme Dihydro orotate dehydrogenase.
- The Eukaryotic enzyme, which contains FMN and Non-heme iron, is located on the outer surface of the inner mitochondrial membrane, where quinines provide its oxidizing power.
Dihydro Orotate + Quinone → Orotate + Reduced Quinone
Step 5: Addition of Ribose-Phosphate Moiety
- Orotate reacts with PRPP to yield Orotidine-5-MonoPhosphate (OMP).
- Orotate phosphoribosyltransferase catalyzes this reaction.
- In this reaction, pyrophosphate is released from the PRPP molecules.
Orotate + PRPP → OMP + PPi
Step 6: Decarboxylation to form UMP
- The final reaction of the pathway is the decarboxylation of OMP by the OMP decarboxylase to form UMP. It is an unusual reaction in that it requires no cofactors.
OMP → UMP + CO2
Synthesis of UTP and CTP
- The synthesis of UTP forms UMP through the phosphate exchange mechanism.
- This reaction is catalyzed by nucleoside monophosphate kinase and Nucleoside diphosphate Kinase.
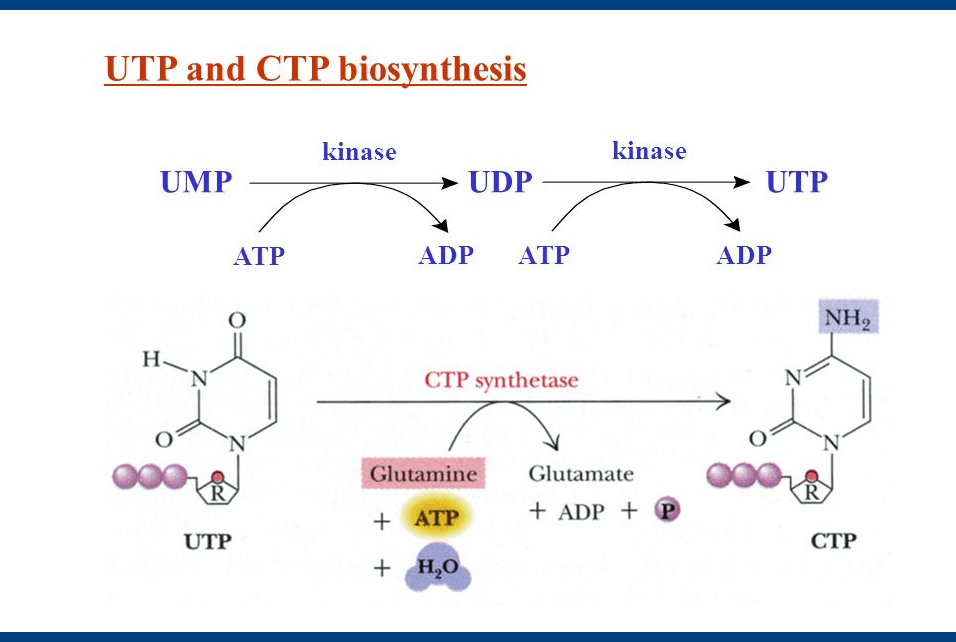
UMP + ATP ↔ UDP + ADP
UDP + ATP ↔ UTP + ADP
The amination of UTP forms CTP by CTP synthetase. In animals, the amino group is donated by Glutamine, whereas it is supplied directly in Ammonia in bacteria.
UTP + Gln +ATP + H2O → CTP + Glu + ADP + Pi
The Synthesis of pyrimidine derivatives is TTP, CTP, and UTP.
Frequently Asked Questions (FAQs) of Pyrimidine Biosynthesis
How are pyrimidines synthesized?
Pyrimidines are synthesized from amines using a series of reactions called the synthesis of purines. The enzyme polynucleotide phosphorylase holoenzyme turns on these reactions. It is an important molecule, as it allows RNA to be converted into nucleic acids that can then serve as a template for synthesizing DNA until later in development when they become functional chromosomes. It makes genes necessary for cell division and the maintenance of closed organelles such as mitochondria in eukaryotic cells that produce energy via ATP synthase.
How many steps are there in pyrimidine synthesis?
There are six steps in pyrimidine synthesis. The growth and division of living organisms require a constant supply of energy. In eukaryotes, it takes place in the form of ATP from adenosine triphosphate (ATP). Phosphorylation is necessary for mitochondrial function, where they can use glucose to produce ATP via oxidative phosphorylation. A free-radical oxidation pathway involves glycolysis, which releases pyruvate to run three different metabolic pathways: the citric acid cycle, pyruvate dehydrogenase (PDH), and Krebs, allowing mitochondria to get energy from glucose by converting it into acetyl-CoA. The acyl group is used to make fatty acids in ketogenesis, powered by oxidizing glucose via oxidative phosphorylation using FAD instead of NAD, which can happen during photosynthesis.
What is the product of pyrimidine synthesis?
Pyrimidine synthesis produces uracil, which is a component of RNA.
What was the first pyrimidine synthesized?
The first pyrimidine synthesized was acetic acid cyanamide.
What is required for pyrimidine ring synthesis?
It is required to have a nitrogen atom in the correct position on the molecule to allow carbon-nitrogen double bonds to form.
Final words on biosynthesis of Pyrimidine
After reading this, you will be able to understand the complete pathway of pyrimidine synthesis, which is a similar process to purine synthesis. You will also understand the structures of the key molecules involved in this process.
You will better understand how purines and pyrimidines are synthesized and why they are essential molecules.
So, whether you are looking to understand how these molecules are synthesized or want to know more about the pathways used in this process, we recommend that you read this blog!
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.